|
Our experts are extremely knowledgeable about gold and can answer all your questions related to the yellow metal. We pride ourselves on being recognised as one of the most reputed precious metal dealers in the country. When you call Physical Gold, your queries need not necessarily be about investments. Find out more about gold from our experts at Physical Gold On the other hand, base metals like iron are more rigid and cannot be bent easily. Gold and silver have more of these planes, making them extremely malleable. 
These are called slip planes, and they allow the metal to bend easily. Metals which have a higher density like gold are more likely to have defects within these planes. This structure is essentially an arrangement of different planes of crystals. Like all other metals, gold has a crystalline structure. The key to our answer lies deep in physics, where we analyse the structure of gold. The softness of a metal is its ability to easily change shape when pressure or stress is applied. If the density of gold is higher than other metals, why is it softer? 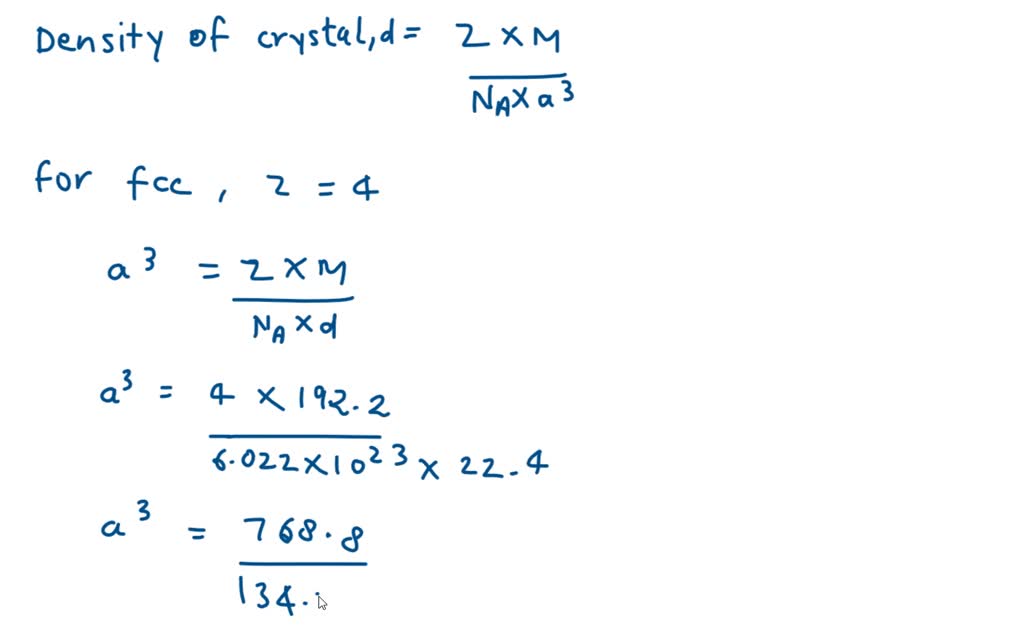
Similarly, a cubic centimetre of silver will weigh 10.5 g Therefore, one cubic centimetre of gold will weigh 19.3 g. So, all we have to do is to weigh a bar of gold and divide this number by its volume. Now, mass is pretty much the same concept as weight. This is the reason why density is represented by grams per cubic centimetre – unit mass per unit volume. Well, the classical definition of the density of an object. The density of silver is approximately half that of gold How can we calculate the density of gold? 
Gold remains unaffected when exposed to air, or most other reagents. Another great property of gold is that it does not corrode easily. This combination of ductility and conductivity make it invaluable in the electronics industry. It is a very good conductor of heat, as well, as electricity. Gold has another property that makes it ideal for several industrial uses. As a ductile metal, it can also be pulled out into wires. The yellow metal is so malleable that 1 ounce can be pressed and spread over an area of 300 square feet. Gold is a highly malleable and ductile metal. In this article, we will briefly overview some of these properties, and also focus on gold density. Have you ever wondered why gold is used widely? Gold has certain properties that ensure its usefulness across industries. Of course, it has numerous other industrial uses. Gold has historically been one of the precious metals of choice for minting coins. The yellow metal is revered around the world for its value and has many different uses. So now further, i can write that this is cube right.Gold and silver are often referred to as the ‘go-to’ precious metals for any investor. Divided by vegeto number is 6.022 multiplying 10 raise to power 23 and density is 22.4 gram per centimeter cube, okay. So this will become a cube, is equal to 4 multiplied by molar. So now i can further write that a cube, that's equal to z, multiplied by m, divided by n, a multiplied by d so now putting the values. It is the volume of unit cell, where a is the adele. Okay, now m is the molar mass and a is the verger number a cube. Z is the number of items per unit cell and for fcc that is face centered cubic unit cell, which is given to us in the question so for this z that is number of atoms per unit cell is 4. So now we have a formula which says that density of unit cell, so that is given by z, multiplied by m, divided by n. So here, first of all, we will find out the edge length of the unit cell with the help of the formula for density of the unit cell and then further we will find out the radius for their respective are length.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
