|
This result is expected to have important implications for the ensemble predictions in many applied fields, and may help to analyze chaotic data sets. The only way is if all species have the same absolute entropy at 0 K. So, for a nonlinear system, though the trajectories of the state variables, say $\ve x$, may appear chaotic in the phase space, say $\Omega$, those of the density function $\rho(\ve x)$ in the new ``phase space'' $L^1(\Omega)$ are not the corresponding Lyapunov exponent is always zero. This shows that the entropy of the two crystalline forms are the same. For rotational entropies, we propose a method based on the variances in Euler angles, which gives good agreement with theory. That is to say, relative entropy is always conserved. Absolute and relative vibrational entropies are found to be better reproduced using quasiharmonic analysis compared to Schlitter's formula. Calculate S at 25C for the reduction of PbO (s),2PbO (s)+C (s)2Pb (s)+CO2 (g) given these absolute entropies:S (J/K.mol)PbO (s),69.45C (s) ,5.7Pb (s),64.89CO2 (s) 213.6+198.8 J/Kmol+353.6JKmol-203.3JKmol+203.3 J/Kmol+488.0JKmol. For a deterministic system, $dH/dt$ is equal to the mathematical expectation of the divergence of the flow (a result obtained before), and, remarkably, $dD/dt = 0$. 
Using standard absolute entropies at 298K, calculate the entropy change for the system when 2.18 moles of H2O (l) react at standard. We conclude that by measuring either the pressure or the energy flux at some known temperature, we can find the absolute entropy of a quantity of thermal. For a stochastic system, its relative entropy $D$ evolves in accordance with the second law of thermodynamics its absolute entropy $H$ may also be so, provided that the stochastic perturbation is additive and the flow of the vector field is nondivergent. Consider the reaction: 2N2 (g) + O2 (g)2N2O (g) Using standard absolute entropies at 298K, calculate the entropy change for the system when 1.91 moles of N2 (g) react at standard conditions. General Chemistry Map: General Chemistry (Petrucci et al.) 19: Spontaneous Change: Entropy and Gibbs Energy 19. San Liang Download PDF Abstract:The evolution of entropy is derived with respect to dynamical systems. Absolute entropy increases steadily with increasing temperature until the melting point is reached, where it jumps suddenly as the substance undergoes a phase. Physical & Theoretical Chemistry Physical Chemistry (LibreTexts) 21: Entropy and the Third Law of Thermodynamics 21. 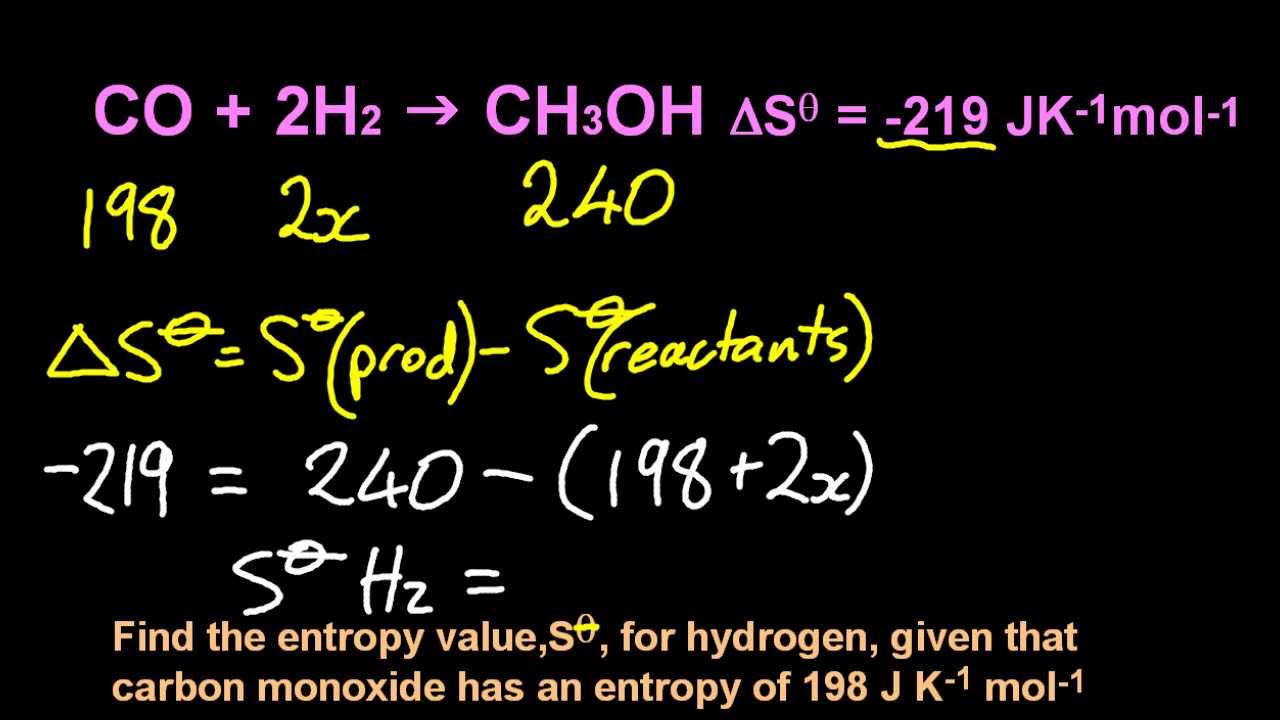
Download a PDF of the paper titled Remarkable evolutionary laws of absolute and relative entropies with dynamical systems, by X.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
